About us
About us
General information
Located in Woippy, Moselle, the Centre d'Études et de Recherche sur l'Appareillage des Handicapés (CERAH) is a department of the Institution Nationale des Invalides (INI), an administrative establishment under the supervision of the Ministry of the Armed Forces.
The CERAH's mission is to conduct research into motor disabilities. It is organised around three units :
- Appliance expertise
- Research and innovation
- Training and documentation
As a place for exchanging ideas, listening, meeting and consulting, our establishment is the interface between professionals and users.
Its medical, scientific and technical expertise in the field of orthopaedic devices, and its particular role as a wheelchair specialist, make it the ideal point of contact for all those involved: disabled people, healthcare professionals, service providers and equipment distributors, researchers, manufacturers, associations, etc.
The CERAH also participates as an expert, as needed, with the High Health Authority and the Economic Committee for Healthcare Products in studying or revising the classification for the reimbursement of assistive products and medical devices by social protection organizations, as well as within various standardization groups of national and international bodies such as AFNOR, CEN, and ISO.
-->The centre has NF EN ISO 9001 certification for all its activities, its laboratories are COFRAC accredited to NF EN ISO 17025 and it has Qualiopi certification for all its training courses.
CERAH Woippy
1, Bellevue BP 50719
57147 WOIPPY CEDEX
Tél. +33 (0)3 87 51 30 30
Fax. +33 (0)3 87 51 30 36

CERAH Antenne de Paris
Institution Nationale des Invalides
6 boulevard des Invalides
CS 70807
75007 PARIS CEDEX 07
Tél. +33 (0)1 40 63 24 63
- Head of CERAH
Joseph Bascou - Deputy Head of CERAH
Martine Delavaquerie - Head of Quality Department
Laure Dentinger
Training and documentation
Objectives
Offer, design and deliver professional training courses in the field of motor disabilities and assistive products.
Receive students at all levels of training.
Monitor medical, social, regulatory, normative and technical developments, and disseminate related information via Cerahtec news published on our home page.
Implement professional training programs designed to :
- become service providers and equipment distributors (PSDM) for disabled people.
- acquire professional skills, particularly in the rental, sale and maintenance of vehicles for the disabled, medical beds and sports equipment for the disabled.
Designing and delivering training courses to meet the needs of potential applicants or changing regulations.
Tutoring students of all levels for their practical training.



The training department holds QUALIOPI certification, attesting to the quality of the training provided.
CERAH has a specialized resource center for the motor-handicapped, whose aim is to build up, process and disseminate its documentary resources.
On the home page of this site, the documentation service provides the latest regulatory and medical information on assistive devices, as well as information on the professions that market, prescribe or dispense these products.
It develops tools, such as thematic dossiers and guides, which it integrates on this site to respond to the various requests of its contacts.
This department is also responsible for carrying out documentary research for users outside the company and/or answering questions, mainly of a regulatory nature.
Appliance expertise
Testing Department
Objectives
To support healthcare manufacturers in their efforts through :
- Technical evaluation of all equipment designed to compensate for motor disability, with a view to CE marking or inclusion on the list of reimbursable products and services.
- Coordination of clinical studies.
Carrying out technical assessments in accordance with French, European and international standards, and with the technical specifications of funding bodies
 Independent and competent laboratory, accredited as a testing laboratory to ISO 17025 for only those tests covered by accreditation n°1-2266.
Independent and competent laboratory, accredited as a testing laboratory to ISO 17025 for only those tests covered by accreditation n°1-2266.
Advice and support in obtaining CE marking and inclusion on the list of reimbursable products and services
Coordination of clinical evaluations throughout the lifecycle of assistive products.
Fittings Department
Objectives
Support patients by providing medico-technical solutions for amputees
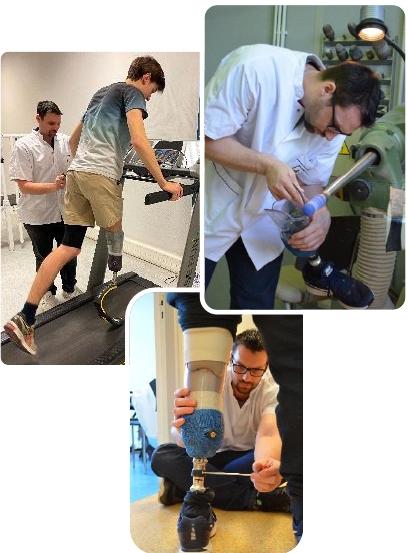
Multidisciplinary consultations
Fitting and follow-up of amputees, war-wounded patients and patients with complex fitting problems
Training healthcare professionals specialized in outpatient orthopedics
Assistive Products and Innovative Clinical Applications Department (PAACI)
Objectives
- Medico-technical expertise
- coordination of clinical studies
- Designing and promoting innovative clinical products
Medico-technical expertise
Multidisciplinary team providing consultations to advise, recommend and prescribe vehicles for the disabled (VPH).
A large fleet of VPHs, and a regularly updated database on Assistance Products, enable this team to provide its medico-technical expertise.


Coordination of clinical studies
A series of tests carried out on humans to demonstrate the medical service rendered by a medical device (MD) and the improvement in quality of life for its users.
Making a medical device accessible to the greatest possible number of people, by assisting manufacturers in the reimbursement process.
Designing and promoting innovative clinical products
Specific projects at the request of disabled people, healthcare professionals or para-sports organizations.
Customized design and adaptation.
Risk analysis and feasibility studies.

Research and Innovation
Objectives
Contribute to the development and improvement of care and quality of life for people with motor disabilities, through the acquisition or creation of :
- New scientific, technical and clinical knowledge
- Scientific and technical tools and methods
- Prototypes for scientific and clinical purposes
Development of scientific knowledge
Goal:
- Prevent the onset of musculoskeletal disorders
- Improve locomotor performance
in wheelchair users, manual wheelchair users and amputees with one or more lower limb amputations.
Methods
- Quantified Motion Analysis, in the laboratory or in the field
- Study and optimization of settings and accessibility

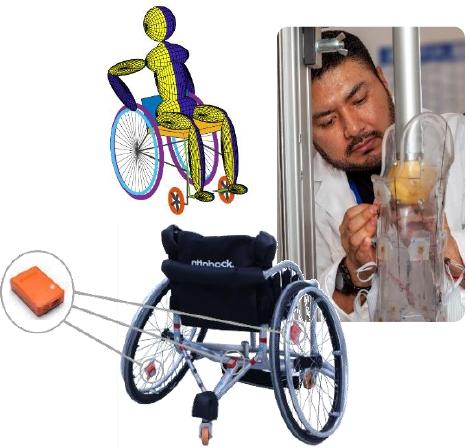
Creation and development of tools and methods
Development of dedicated software for research and clinical practice
Experimental protocols and specific data processing algorithms
Creation of specific equipment
Prototype design
Mechanics and electronics
Test benches and specific sensors
Specific instrumented ergometers

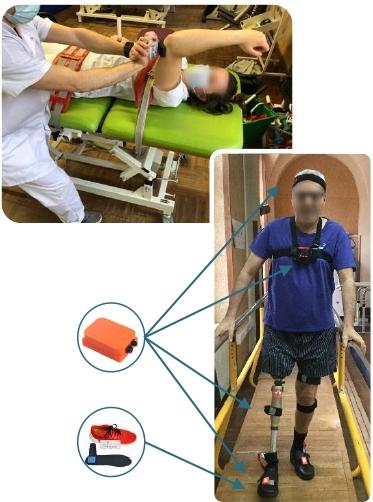
Transfers to the clinic
Supervision of scientific projects for medical staff
Evaluation of rehabilitation protocols
Study of the contribution of connected objects in rehabilitation
Methodological support for clinical research
The Research and Innovation unit is made up of researchers, engineers and technicians specializing in mechanics, electronics and IT.


